|
 |
| Encephalitis > Volume 3(2); 2023 > Article |
|
Abstract
Notes
Conflicts of Interest
Jangsup Moon has been an associate editor of encephalitis since October 2020. Sang Kun Lee and Kon Chu have been the editorial boards of encephalitis since October 2020. They were not involved in the review process of this original article. No other potential conflict of interest relevant to this article was reported.
Author Contributions
Conceptualization: Chu K; Formal analysis, Project administration: Moon J; Visualization: Lee Y, Lee HS, Kim YM, Lee S, Park HY; Investigation: Kim Y, Lee S; Software: Ahn SJ; Supervision: Ahn SJ, Lee SK, Chu K; Validation: Lee HS, Park H, Lee SK; Writing–original draft: Lee Y, Moon J; Writing–review & editing: Ahn SJ, Lee SK, Chu K.
Figure 1.
Timeline of clinical course and treatment of the patient
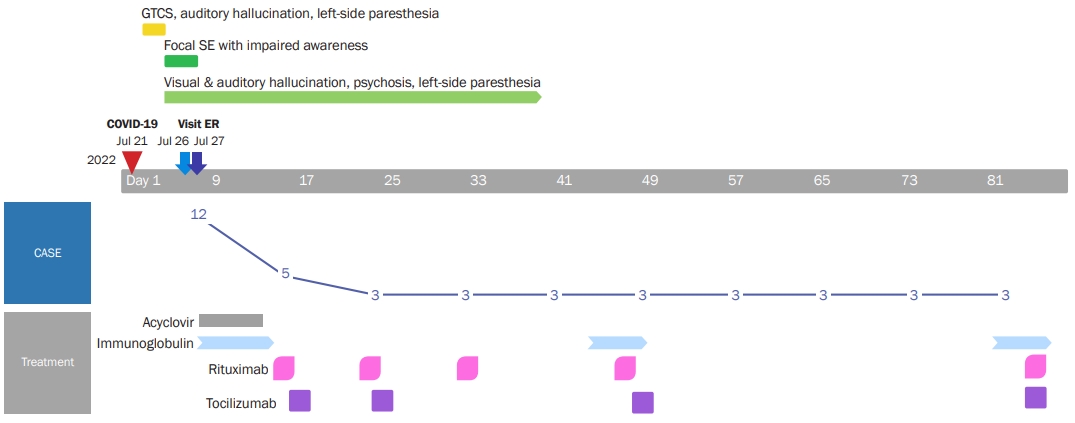
Figure 2.
Brain MRI and brain SPECT findings
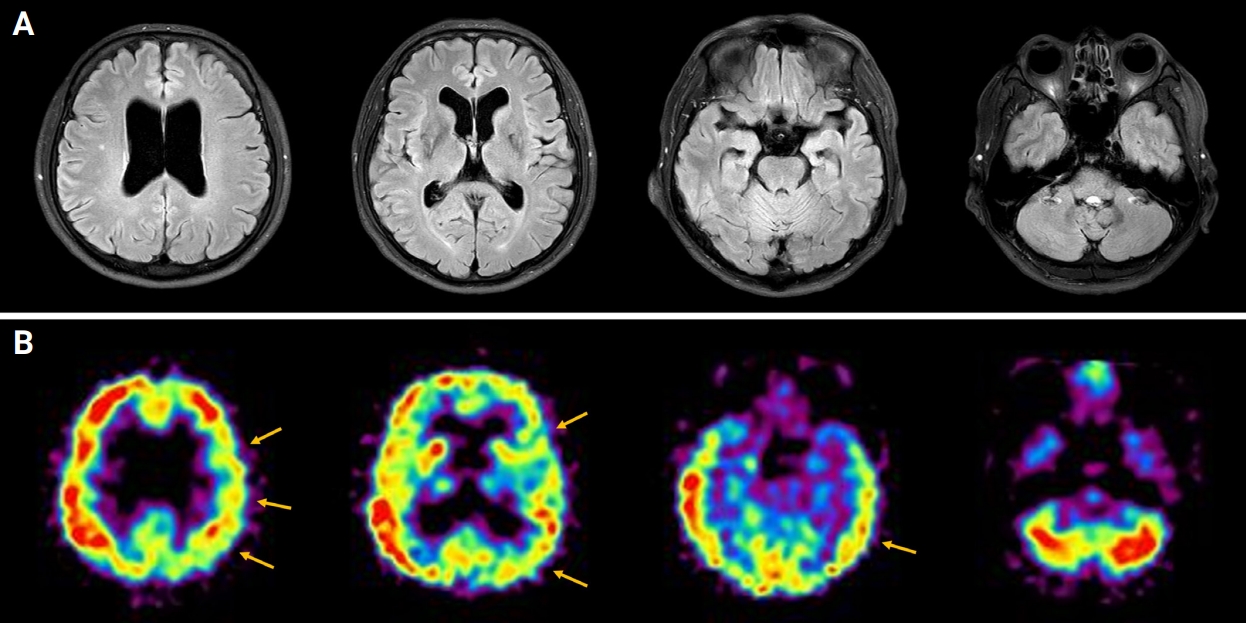
Figure 3.
EEG findings
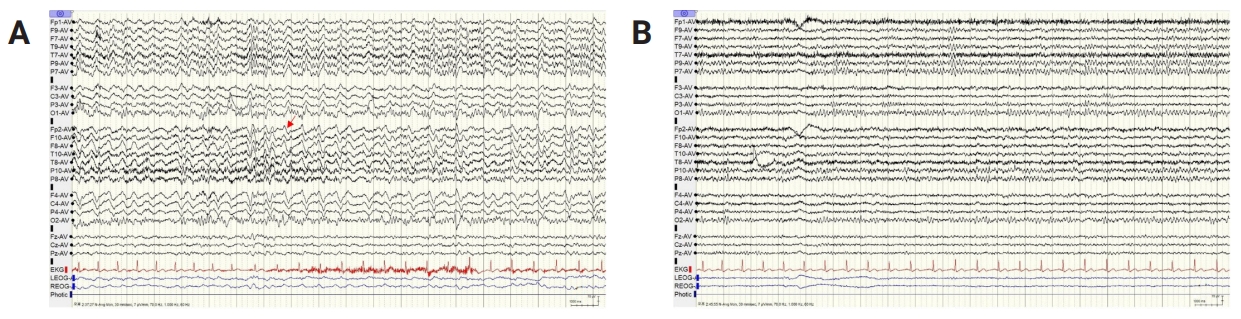
Table 1
| Study | Year | Age (yr)/sex | Interval from COVID-19 to onset (day) | MOG antibody titer | Neurologic syndrome | SARS-CoV-2 test | CSF WBC | Treatment |
|---|---|---|---|---|---|---|---|---|
| Ide et al. [12] | 2022 | 24/F | 14 | Unknown | ADEM, ON | Nasopharyngeal (+) | WBC 16 cells/µL | Methylprednisolone, tapered to oral prednisolone |
| CSF (–) | ||||||||
| Johnsson et al. [10] | 2022 | 25/F | 14 | 1:1,000 positive | ADEM | Nasopharyngeal (+) | Lymphocytes 295 × 106/L, | Methylprednisolone, tapered to oral prednisolone, plasmapheresis, methotrexate |
| CSF (–) | neutrophil 229 × 106/L | |||||||
| 20/M | 56 | 1:100 positive | ON, myelitis | Nasopharyngeal (+) | Lymphocytes 195 × 106/L | Plasmapheresis, methylprednisolone, tapered to oral prednisolone | ||
| 29/M | 14 | 1:100 positive | Bilateral ON | Nasopharyngeal (+) | NA | Methylprednisolone, tapered to oral prednisolone | ||
| 60/F | 54 | 1:1,000 positive | Unilateral ON | Nasopharyngeal (+) | NA | Methylprednisolone, tapered to oral prednisolone | ||
| Yang et al. [13] | 2022 | 57/M | 21 | 1:100 positive | Myelitis | Nasopharyngeal (+) | WBC 9/µL | Methylprednisolone, tapered to oral prednisolone |
| Colantonio et al. [14] | 2022 | 41/M | 16 | 1:100 positive | ADEM, ON | Nasopharyngeal (+) | WBC 45/µL | Methylprednisolone, plasmapheresis |
| Durovic et al. [15] | 2021 | 22/F | 10 | 1:640 positive | Encephalitis | Nasopharyngeal (+) | WBC 31/µL | Methylprednisolone |
| CSF (–) | ||||||||
| Kogure et al. [16] | 2021 | 47/M | 2 | 1:128 positive | Bilateral ON | Nasopharyngeal (+) | NA | Methylprednisolone, tapered to oral prednisolone |
| Peters et al. [17] | 2021 | 23/M | 35 | 1:100 positive | Encephalitis | Nasopharyngeal (+) | WBC 1/µL (WBC 57/µL at repeated study) | Methylprednisolone |
| CSF (–) | ||||||||
| Rojas-Correa et al. [18] | 2021 | 69/M | 45 | Unknown | Bilateral ON | Nasopharyngeal (+) CSF (–) | WBC 2/µL | Methylprednisolone, tapered to oral prednisolone |
| Sawalha et al. [19] | 2020 | 44/M | 14 | 1:160 positive | Bilateral ON | Nasopharyngeal (+) | WBC 3/µL | Methylprednisolone, tapered to oral prednisolone |
| Woodhall et al. [11] | 2020 | 39/F | 6 | 1:800 positive | MOGAD relapse; ON | Nasopharyngeal (+) | NA | Mycophenolate mofetil, prednisolone |
| Pinto et al. [20] | 2020 | 44/F | 7 | Unknown | CNS vasculopathy | Nasopharyngeal (+) | WBC 13/µL | Methylprednisolone, tapered to oral prednisolone, plasmapheresis |
MOG, myelin oligodendrocyte glycoprotein; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; COVID-19, coronavirus disease 2019; CSF cerebrospinal fluid; WBC, white blood cells; F, female; M, male; ADEM, acute disseminated encephalomyelitis; ON, optic neuritis; NA, not applicable; MOGAD, MOG-immunoglobulin G disease; CNS, central nervous system.




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print